
Nucleocytoplasmic communication
and the role of the nuclear pore
complex |
|
|
| Macromolecular communication between the nucleus
and the cytoplasm is one of the hallmarks of the eukaryotic cell. It
connects nuclear and cytoplasmic processes such as transcription and
translation and introduces a level of regulation that is both rapid and
easily reversible.
Transport of RNAs and proteins and between the
nucleus and cytoplasm occurs through large proteinaceous structures that
are embedded in the nuclear envelope, called nuclear pore complexes
(NPCs). These 125 MDa complexes restrict free diffusion of molecules or
particles with a diameter larger than 9 nm, corresponding to the size of a
globular protein of 40-60 kD. However, complexes of up to 25 nm in
diameter (several megadaltons) are efficiently transported provided they
carry a nuclear import or export signal. |

The nuclear
pore complex |
| In both lower and higher eukaryotes, the NPC is
made up of 30-50 different proteins that are collectively named nucleoporins,
and occupy distinct positions within the NPC. Although many specific
transport routes upstream of the NPC have been elucidated, specificity
within the nuclear pore complex remains largely unknown.
Nucleocytoplasmic transport is conducted by
distinct classes of soluble transport receptors that interact with both
cargo and nuclear pore complex. One class consists of importins and
exportins,
a superfamily of transport receptors that are responsible for the majority
of nuclear transport pathways identified to date. Importins mediate import
of several different classes of (ribonucleo) proteins, while exportins
mediate nuclear exit of proteins, tRNAs and U snRNAs.
Importin and exportin mediated transport is
dependent on a signal within the transport substrate. Examples of import
signals are the basic nuclear localization signal (NLS) that is found in
many nuclear proteins. Similarly, the nuclear export signal (NES) mediates
rapid nuclear export. Leucine-rich-type NESs have recently been identified
in a diverse range of cellular proteins that include cell cycle
regulators, stress response signal transducers and mediators of apoptosis.
There are at least nine different importins and
five exportins,
each recognizing a different or a different subset of transport
signals.
|
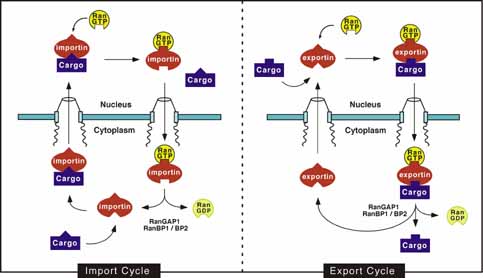
Importin and exportin mediated nucleocytoplasmic
transport |
| The small Ras-like GTPase Ran in the GTP-bound
form (RanGTP) imposes directionality to importin and exportin mediated
transport. RanGTP is only present in high concentrations in the nucleus
where it dissociates importin-cargo complexes and stabilizes
exportin-cargo complexes (see Figure 2). The RanGTP gradient across the
nuclear envelope is maintained by nuclear localization of its exchange
factor, RCC1, and cytoplasmic localization of its GTPase activating
enzyme, RanGAP. |
|
Past research
In the lab Gerard Grosveld (St. Jude's Children's
Research Hospital, Memphis, USA), we identified proteins that interacted
with the nucleoporin CAN/Nup214. One of them was the human homologue of a
yeast protein named CRM1, that we proposed to be a new transport factor.
This proposal was based on experimental data, and on sequence homology
with the import receptor importin b. We
identified a group of related proteins with homology to both CRM1 and
importin b that was proposed new family of
transport factors (Fornerod et al., 1997a).
In the lab of Iain Mattaj (EMBL, Heidelberg,
Germany), together with Mutsuhito Ohno, we established that CRM1 was an
export receptor for "leucine rich" nuclear export signals (see above). We
showed that CRM1 binds directly to the export signal, and that this can be
inhibited by the fungal cytotoxin leptomycin B (LMB). Leptomycin B was
also shown to directly bind to CRM1. NES binding was only stable upon
cooperative binding of the small GTPase Ran in its GTP-bound form. This
suggested the mechanism with which the RanGTP gradient across the nuclear
envelope directs transport of NES-containing (ribonucleo) proteins, and we
proposed that this mechanism would be more generally applicable to nuclear
export processes (Fornerod et al., 1997b). |
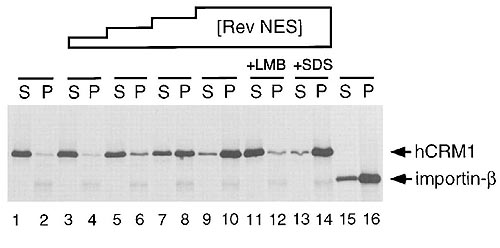
Pull-down assay on RanGTP beads showing that CRM1
binding to the HIV-1 Rev NES
is RanGTP-dependent and
leptomycin B sensitive. S, supernatant; P, pellet. |
| Indeed, we could show that a trimeric
CRM1-NES-RanGTP complex is disassembled by Ran cofactors that are present
at the cytoplasmic side of the NPC. With a quantitative CRM1/NES-cargo
binding assay, we demonstrated that there are significant differences
among natural NESs in affinity for CRM1, suggesting that the steady state
nucleo-cytoplasmic distribution of a shuttling protein could be determined
by the relative strength of its NES (Askjaer et al., 1999).
With Gert-Jan Arts, post-doc in Iain Mattaj's lab,
we identified the nuclear export receptor for tRNAs, exportin-t, another
member of the importin b family. Similar to CRM1,
exportin-t bound directly to its tRNA cargo in a RanGTP-dependent manner
(Arts et al., 1998).
Refocussing on the nuclear pore complex, we have
extended the analysis of the CAN/CRM1 interaction, and demonstrated that
the interaction dependents on RanGTP, suggesting a role of this
interaction in export complex disassembly and CRM1 reimport (Askjaer et
al., 1999). |
|
Present Research
It has recently become clear that many cellular
processes, such as signal transduction, cell cycle progression and
apoptosis are regulated on the level of nuclear transport. Much of this
progress was based on the identification of transport signals, transport
receptors, and understanding of mechanisms of the directionality of
transport.
Such basic understanding is clearly lacking of the
role of the NPC in nucleocytoplasmic transport. Therefore we study the
basic function of nuclear pore complex in nucleocytoplasmic transport
pathways. In parallel, we are exploring to what extend the NPC functions
as a regulator of transport and as a regulator of other cellular
processes.
We are using a combination of biochemistry and
cell biology to understand the mechanism of nucleocytoplasmic transport
and NPC function. Biochemical methods can define transport interactions on
a molecular level, while functional cell biological assays are necessary
to address the role of the nuclear transport interactions.
One of the experimental systems used is nuclear
reconstitution. Egg extracts of the african clawed frog, Xenopus
laevis, support efficient nuclear formation around demembrenated sperm
chromatin in vitro. These nuclei contain numerous nuclear pore
complexes, and actively import protein substrates. Also the DNA can
undergo one round of replication.
Nuclear transport can be monitored by accumulation
of exogenously added fluorescently labelled substrate (see Figure below).
Mutant NPCs can be created by removing NPC components from the egg extract
prior to nuclear assembly. Those mutant NPCs can be studied for transport
and other defects. Add back of purified or recombinant nucleoporins can
demonstrate specificity, and is used to identify regions in the protein
responsible for the observed effects. |

Fluorescently labelled BSA-NLS imported into in vitro reconstituted
nuclei. |
| Other methods that are used to study nuclear
transport and NPC function include nuclear and cytoplasmic microinjection
of labelled proteins and RNAs (see below). |
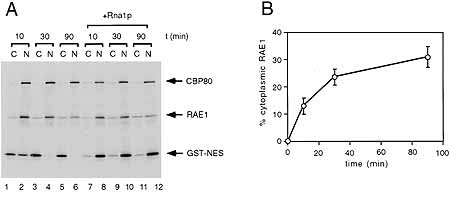
Example of a nuclear export assay. A mixture of labelled proteins
is microinjected into Xenopus oocyte nuclei,
and
appearance in the cytoplasm is monitored in time by
gelelectrophoresis(panel A) of cytoplasmic (C) and
nuclear (N) fractions. Panel B: quantitation of cytoplasmic
accumulation of RAE1. |
|
Research Opportunities
Undergratuate student projects are usually
available. In addition Ph.D and postdoctoral positions may be available;
please inquire. |
|
Contact
Maarten Fornerod fornerod@nki.nl
Netherlands Cancer Institute - H4
Room 416/ Lab 419
Plesmanlaan 121 - 1066 CX Amsterdam, The Netherlands
Tel: +31-(0)20-512 2024 |
|
Selected publications
Maarten Fornerod and Mustuhito Ohno (2002). Exportin-mediated nuclear
export of proteins and ribonuleoproteins. Res. Probl. Cell Diff.
35:67-91 (in press).
Tobias C. Walther, Maarten Fornerod, Helen Pickersgill, Martin
Goldberg, Terry D. Allen and Iain W. Mattaj (2001). The Nucleoporin Nup153
is Requred for Nuclear Pore Basket Formation, Nuclear pore Complex
Anchoring and Import of a Subset of Nuclear Proteins. EMBO J.
20:5703-5714.
Ludwig Englmeier, Maarten Fornerod, F. Ralf Bischoff, Carlo Petosa,
Iain W. Mattaj and Ulrike Kutay (2001). RanBP3 facilitates nuclear protein
export by stabilising the interaction between certain export substrates
and CRM1. EMBO Rep. 2:926-932.
Jörg Hamm and Maarten Fornerod (2000). Anti-idiotype RNAs that mimic
the leucine-rich nuclear export signal and specifically bind to
CRM1/exportin 1. Chem. Biol. 7:345-54.
Peter Askjaer, Angela Bachi, Matthias Wilm, Ralf Bischoff, Daniel
Weeks, Vera Ogniewski, Mutsuhito Ohno, Christof Niehrs, Jorgen Kjems, Iain
W. Mattaj and Maarten Fornerod (1999). RanGTP-regulated interactions of
CRM1 with nucleoporins and a shuttling DEAD-box helicase. Mol. Cell.
Biol. 19: 6276-6285.
Colin Pritchard, Fornerod M., Lawryn L. Kasper, and Jan M. van Deursen.
(1999). RAE1 is a shuttling mRNA export factor that binds to a GLEBS-like
NUP98 motif at the nuclear pore complex through multiple domains. J.
Cell Biol. 145:237-54.
Gert-Jan Arts, Maarten Fornerod and Iain W. Mattaj (1998).
Identification of a nuclear export receptor for tRNA. Curr. Biol.
8:305-14.
Mutsohito Ohno, Maarten Fornerod and Iain W. Mattaj (1998).
Nucleocytoplasmic transport: the last 200 nanometers. Cell
92:327-36.
Maarten Fornerod, Mutsohito Ohno, Mironu Yoshida and Iain W. Mattaj
(1997b). CRM1 is a nuclear export receptor for leucine-rich nuclear export
signals. Cell 90: 1051-1060.
Maarten Fornerod, Jan van Deursen, Sjozèf van Baal, Donna Davis, K.
Gopal Murti and Gerard Grosveld (1997a). The human homologue of yeast CRM1
is in a dynamic subcomplex with CAN/Nup214 and a novel nuclear pore
component Nup88. EMBO J. 16: 807-816. |
|
 |
 |
|